Bioventus werft eerste patiënt voor BONES, onderzoek naar EXOGEN®
DURHAM, N.C.–(BUSINESS WIRE)– Bioventus, internationaal toonaangevend in orthobiologische middelen, heeft vandaag bekendgemaakt dat de eerste patiënten zich hebben aangemeld voor het klinische ontwikkelingsprogramma BONES (Bioventus Observational Non-interventional EXOGEN Studies). BONES bestaat uit drie niet-interventionele observatieonderzoeken in direct contact met patiënten. Het onderzoek is opgezet om praktische gegevens te verzamelen over het gebruik van het EXOGEN Ultrasound Bone Healing System. EXOGEN maakt gebruik van veilige, effectieve, laag-intensieve, gepulseerde ultrageluidtherapie (LIPUS) ter stimulatie van het natuurlijke genezingsproces van botten.
Bioventus Announces First Patients Enrolled in BONES Study for EXOGEN® |
|||||
|
DURHAM, N.C.–(BUSINESS WIRE)– Bioventus, a global leader in orthobiologics, today announced the first patients have been enrolled in its BONES (Bioventus Observational Non-interventional EXOGEN Studies) clinical development program. BONES includes three observational, non-interventional, direct-to-patient studies designed to collect real world data on the use of the EXOGEN Ultrasound Bone Healing System. EXOGEN uses safe, effective low-intensity pulsed ultrasound (LIPUS) to help stimulate the body’s natural bone healing process. This Smart News Release features multimedia. View the full release here: http://www.businesswire.com/news/home/20170913005740/en/ 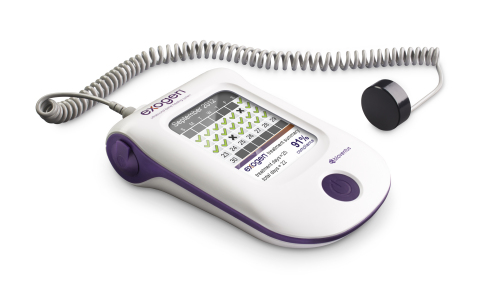 EXOGEN Ultrasound Bone Healing System from Bioventus (Photo: Business Wire) EXOGEN Ultrasound Bone Healing System from Bioventus (Photo: Business Wire)
The BONES studies will compare the incidence of fracture nonunions in patients utilizing EXOGENwith patients from a national health insurance claims database who received standard of care alone. The studies will include long and small bones of upper and lower extremities and the unique design of BONES was discussed with FDA during its development. Its primary endpoint is defined as the ability of EXOGEN to mitigate the risk of a fracture progressing to nonunion in the presence of known risk factors. “We are excited to build on the vast clinical experience available on this established therapy and further develop our knowledge on the real world effectiveness of EXOGEN, by assessing its value in reducing nonunions, a highly disabling complication that also carries a significant societal burden,” said Alessandra Pavesio, Senior Vice President and Chief Science Officer, Bioventus. “Accomplishing this ambitious goal requires an alternative methodology to randomized controlled trials and involves epidemiologically grounded research of large patient cohorts enrolled both prospectively, and within the big data available from US health insurance claims.” About Bioventus Bioventus is an orthobiologics company that delivers clinically proven, cost-effective products that help people heal quickly and safely. Its mission is to make a difference by helping patients resume and enjoy active lives. The company has two product portfolios for orthobiologics, Bioventus Active Healing Therapies and Bioventus Surgical that make it a global leader in active orthopaedic healing. Its EXOGEN® Ultrasound Bone Healing System uses safe, effective low intensity pulsed ultrasound (LIPUS) to stimulate the body’s natural healing process. EXOGEN has been used to treat more than 1 million patients worldwide and numerous regulatory agencies including the FDA, Health Canada, BSI, TGA, Medsafe, UAE Ministry of Health and SFDA have granted their approval of the product. Today it is the leading bone healing system in the market with complaints for lack of efficacy averaging less than 1%. Built on a commitment to high quality standards, evidence-based medicine and strong ethical behavior, Bioventus is a trusted partner for physicians worldwide. For more information, visit www.BioventusGlobal.com and follow the company on Twitter @Bioventusglobal. Bioventus, the Bioventus logo, and EXOGEN are registered trademarks of Bioventus LLC. EXOGEN – Summary of Indications for Use in the US *Summary of Indications for Use: The EXOGEN Ultrasound Bone Healing System is indicated for the non-invasive treatment of established non-unions* excluding skull and vertebra. In addition, EXOGEN is indicated for accelerating the time to a healed fracture for fresh, closed, posteriorly displaced distal radius fractures and fresh, closed or Grade I open tibial diaphysis fractures in skeletally mature individuals when these fractures are orthopaedically managed by closed reduction and cast immobilization. There are no known contraindications for the EXOGEN device. Safety and effectiveness has not been established for individuals lacking skeletal maturity; pregnant or nursing women; patients with cardiac pacemakers; on fractures due to bone cancer; or on patients with poor blood circulation or clotting problems. Some patients may be sensitive to the ultrasound gel. Full prescribing information can be found in product labeling, at www.exogen.com or by contacting customer service at 1-800-836-4080. *A nonunion is considered to be established when the fracture site shows no visibly progressive signs of healing. View source version on businesswire.com: http://www.businesswire.com/news/home/20170913005740/en/ Contacts Bioventus |
|||||
