HistoSonics Notches Significant Reimbursement Wins
Company Awarded Increased Payment Rate for Liver Procedure and New Unique CPT® Code for its Kidney Application
MINNEAPOLIS–(BUSINESS WIRE)– HistoSonics, (www.histosonics.com), the manufacturer of the non-invasive Edison® Histotripsy System and novel sonic beam therapy, announced today that the Centers of Medicare & Medicaid Services (CMS) set a new payment rate for histotripsy of liver tumors procedures, increasing the payment rate to $17,500. In their November 2023 rule making process, CMS utilized claims data from the #HOPE4LIVER Trial conducted in the United States to increase the outpatient payment rate for HistoSonics Category III Current Procedural Terminology (CPT®) Code 0686T, the non-thermal ablation via acoustic energy delivery of malignant hepatocellular tissue including image guidance, from $12,500 to the new national average rate of $17,500, assigning the novel procedure to Ambulatory Payment Classification (APC) 1576. This payment rate supports hospitals and physicians using the non-invasive histotripsy procedure in the outpatient setting to destroy targeted liver tumors while existing Diagnosis Related Group (DRG) payment rates are applicable for patients who require hospital admission.
This press release features multimedia. View the full release here: https://www.businesswire.com/news/home/20231106087108/en/
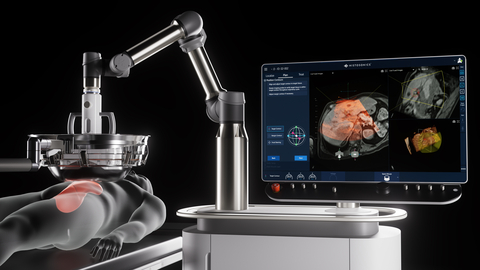
HistoSonics Edison Histotripsy System (Photo: Business Wire)
“CMS’s reassignment of the histotripsy liver procedure payment rate enables Medicare patients much needed access to histotripsy and its potential benefits. We commend CMS on facilitating access to breakthrough devices like ours for Medicare patients, many of whom historically have had delayed access to innovative technology,” commented HistoSonics President and CEO Mike Blue. HistoSonics’ Edison Histotripsy System combines advanced imaging and proprietary software to deliver patient specific treatments and uses the science of histotripsy to mechanically destroy targeted tissues at sub-cellular levels. Histotripsy has been shown to be safe and effective in destroying targeted liver tumors, including unresectable liver tumors, recently gaining clearance from the FDA to market the Edison System for the non-invasive destruction of liver tumors, using a non-thermal, mechanical process of focused ultrasound.
Additionally, the American Medical Association (AMA) has recently issued a new Category III CPT code for histotripsy of kidney tissue. This is significant as it provides patients and clinicians with the first automated external beam therapy using acoustic energy to mechanically destroy diseased kidney tissue without using incisions, ionizing radiation, or heat. The new CPT code will become effective on July 1, 2024, in the US at which time providers can utilize the code when performing histotripsy procedures on patients across the United States as part of the company’s sponsored Investigational Device Exemption (IDE) trial named the “#HOPE4KIDNEY Trial”.
CPT codes are granted and regulated by the AMA CPT® Editorial Panel and are widely used by government payers, including Medicare and Medicaid, and commercial health plans to describe healthcare services and procedures for reimbursement. The release of this new code represents a major reimbursement milestone as hospitals across the US will be able to submit claims directly related to the service. Upon FDA marketing authorization for histotripsy of the kidney this new code will provide patients and physicians access to histotripsy as an option to treat kidney tissue non-invasively and without potential complications seen with conventional therapies like bleeding, infection, or pain from surgery.
The new Category III CPT code, X201T Histotripsy (i.e., non-thermal ablation via acoustic energy delivery) of renal tissue, will be released January 1, 2024.
“Obtaining this kidney CPT code is another significant milestone in our progress to bring histotripsy to the clinic, enabling providers with a mechanism to capture data on the use of histotripsy for destroying targeted kidney tissue and will facilitate reimbursement with payers to enable broad access for the many patients who could potentially benefit from histotripsy’s unique capabilities,” added Blue.
The Edison System is investigational for use in the kidney and is only available for sale in the United States for its intended use in destroying liver tumors.
About HistoSonics
HistoSonics is a privately held medical device company developing non-invasive platforms and proprietary sonic beam therapy utilizing the science of histotripsy, a novel mechanism of action that uses focused ultrasound to mechanically destroy and liquify unwanted tissue and tumors. The company is currently focused on commercializing their Edison System in the US and select global markets for liver treatment while expanding histotripsy applications into other organs like kidney, pancreas, and others. HistoSonics has offices in Ann Arbor, Michigan and Minneapolis, MN.
For more information please visit: www.histosonics.com/
View source version on businesswire.com: https://www.businesswire.com/news/home/20231106087108/en/
Contacts
Media contact:
Josh King
Vice President of Global Market Access
Email : Joshua.king@histosonics.com
Phone : +1 608.332.8124

